in this temperature range, what is the percentage change in resistance due to thermal expansion?
Temperature and Rut
iv Thermal Expansion
Learning Objectives
By the stop of this department, you will be able to:
- Answer qualitative questions about the effects of thermal expansion
- Solve problems involving thermal expansion, including those involving thermal stress
The expansion of alcohol in a thermometer is ane of many commonly encountered examples of thermal expansion, which is the change in size or volume of a given organization every bit its temperature changes. The well-nigh visible example is the expansion of hot air. When air is heated, information technology expands and becomes less dense than the surrounding air, which then exerts an (up) force on the hot air and makes steam and smoke rise, hot air balloons bladder, and and so forth. The aforementioned behavior happens in all liquids and gases, driving natural heat transfer upwardly in homes, oceans, and atmospheric condition systems, as we volition discuss in an upcoming section. Solids too undergo thermal expansion. Railroad tracks and bridges, for example, accept expansion joints to allow them to freely expand and contract with temperature changes, equally shown in (Figure).
(a) Thermal expansion joints like these in the (b) Auckland Harbour Span in New Zealand allow bridges to change length without buckling. (credit: modification of works by "ŠJů"/Wikimedia Eatables)

What is the underlying crusade of thermal expansion? As previously mentioned, an increase in temperature means an increase in the kinetic free energy of individual atoms. In a solid, unlike in a gas, the molecules are held in place by forces from neighboring molecules; every bit nosotros saw in Oscillations, the forces can be modeled as in harmonic springs described by the Lennard-Jones potential. Energy in Uncomplicated Harmonic Motion shows that such potentials are asymmetrical in that the potential energy increases more steeply when the molecules get closer to each other than when they get further away. Thus, at a given kinetic energy, the distance moved is greater when neighbors move away from each other than when they move toward each other. The result is that increased kinetic free energy (increased temperature) increases the average distance betwixt molecules—the substance expands.
For almost substances under ordinary conditions, information technology is an excellent approximation that at that place is no preferred direction (that is, the solid is "isotropic"), and an increase in temperature increases the solid's size past a sure fraction in each dimension. Therefore, if the solid is free to expand or contract, its proportions stay the same; only its overall size changes.
Linear Thermal Expansion
Co-ordinate to experiments, the dependence of thermal expansion on temperature, substance, and original length is summarized in the equation
![]()
where ![]() is the alter in length
is the alter in length ![]() is the change in temperature, and
is the change in temperature, and ![]() is the coefficient of linear expansion, a material property that varies slightly with temperature. As
is the coefficient of linear expansion, a material property that varies slightly with temperature. As ![]() is nearly constant and also very small, for practical purposes, we apply the linear approximation:
is nearly constant and also very small, for practical purposes, we apply the linear approximation:
![]()
(Figure) lists representative values of the coefficient of linear expansion. Equally noted before, ![]() is the aforementioned whether it is expressed in units of degrees Celsius or kelvins; thus,
is the aforementioned whether it is expressed in units of degrees Celsius or kelvins; thus, ![]() may have units of
may have units of ![]() or 1/G with the same value in either case. Approximating
or 1/G with the same value in either case. Approximating ![]() as a constant is quite accurate for small changes in temperature and sufficient for most practical purposes, even for big changes in temperature. We examine this approximation more closely in the next example.
as a constant is quite accurate for small changes in temperature and sufficient for most practical purposes, even for big changes in temperature. We examine this approximation more closely in the next example.
| Cloth | Coefficient of Linear Expansion | Coefficient of Volume Expansion |
|---|---|---|
| Solids | ||
| Aluminum | | |
| Brass | | |
| Copper | | |
| Gold | | |
| Iron or steel | | |
| Invar (nickel-iron blend) | | |
| Atomic number 82 | | |
| Silvery | | |
| Glass (ordinary) | | |
| Drinking glass (Pyrex®) | | |
| Quartz | | |
| Concrete, brick | | |
| Marble (average) | | |
| Liquids | ||
| Ether | | |
| Ethyl alcohol | | |
| Gasoline | | |
| Glycerin | | |
| Mercury | | |
| H2o | | |
| Gases | ||
| Air and most other gases at atmospheric pressure | | |
Thermal expansion is exploited in the bimetallic strip ((Figure)). This device tin can be used as a thermometer if the curving strip is attached to a pointer on a scale. It tin can also be used to automatically shut or open up a switch at a certain temperature, as in older or analog thermostats.
Calculating Linear Thermal Expansion The primary span of San Francisco'due south Golden Gate Bridge is 1275 thousand long at its coldest. The span is exposed to temperatures ranging from ![]() to
to ![]() . What is its change in length betwixt these temperatures? Assume that the bridge is made entirely of steel.
. What is its change in length betwixt these temperatures? Assume that the bridge is made entirely of steel.
Strategy Use the equation for linear thermal expansion ![]() to calculate the modify in length,
to calculate the modify in length, ![]() . Use the coefficient of linear expansion
. Use the coefficient of linear expansion ![]() for steel from (Figure), and note that the change in temperature
for steel from (Figure), and note that the change in temperature ![]() is
is ![]()
Solution Substitute all of the known values into the equation to solve for ![]() :
:
![]()
Significance Although non large compared with the length of the bridge, this modify in length is observable. It is generally spread over many expansion joints so that the expansion at each joint is small.
Thermal Expansion in Two and Three Dimensions
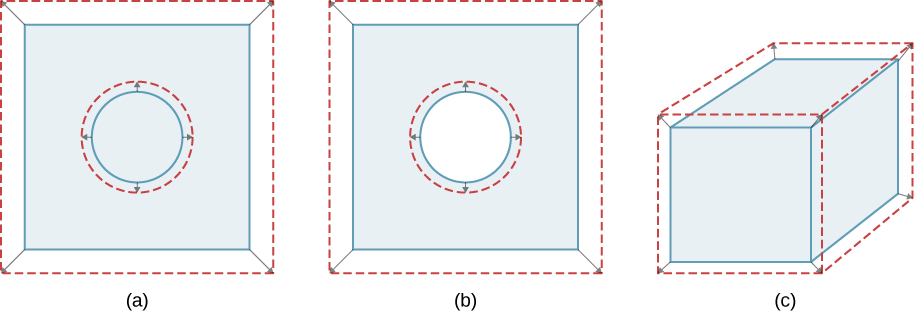
Unconstrained objects expand in all dimensions, as illustrated in (Figure). That is, their areas and volumes, too as their lengths, increment with temperature. Because the proportions stay the aforementioned, holes and container volumes also get larger with temperature. If you cutting a hole in a metal plate, the remaining material volition expand exactly as it would if the piece yous removed were all the same in place. The piece would get bigger, so the hole must get bigger too.
Thermal Expansion in 2 Dimensions
For small temperature changes, the modify in area ![]() is given by
is given by
![]()
where ![]() is the alter in area
is the alter in area ![]() is the change in temperature, and
is the change in temperature, and ![]() is the coefficient of linear expansion, which varies slightly with temperature. (The derivation of this equation is analogous to that of the more than of import equation for three dimensions, below.)
is the coefficient of linear expansion, which varies slightly with temperature. (The derivation of this equation is analogous to that of the more than of import equation for three dimensions, below.)
In general, objects expand in all directions every bit temperature increases. In these drawings, the original boundaries of the objects are shown with solid lines, and the expanded boundaries with dashed lines. (a) Expanse increases because both length and width increase. The surface area of a round plug also increases. (b) If the plug is removed, the hole it leaves becomes larger with increasing temperature, just as if the expanding plug were all the same in place. (c) Book likewise increases, because all three dimensions increase.
Thermal Expansion in Three Dimensions
The human relationship between volume and temperature ![]() is given by
is given by ![]() , where
, where ![]() is the coefficient of volume expansion. As you tin bear witness in (Figure),
is the coefficient of volume expansion. As you tin bear witness in (Figure), ![]() . This equation is usually written equally
. This equation is usually written equally
![]()
Note that the values of ![]() in (Effigy) are equal to
in (Effigy) are equal to ![]() except for rounding.
except for rounding.
Book expansion is divers for liquids, but linear and expanse expansion are not, equally a liquid'south changes in linear dimensions and area depend on the shape of its container. Thus, (Figure) shows liquids' values of ![]() merely non
merely non ![]() .
.
In full general, objects expand with increasing temperature. H2o is the most important exception to this rule. Water does aggrandize with increasing temperature (its density decreases) at temperatures greater than ![]() . Yet, it is densest at
. Yet, it is densest at ![]() and expands with decreasing temperature between
and expands with decreasing temperature between ![]() and
and ![]() (
(![]() ), as shown in (Figure). A striking effect of this phenomenon is the freezing of water in a pond. When water near the surface cools downward to
), as shown in (Figure). A striking effect of this phenomenon is the freezing of water in a pond. When water near the surface cools downward to ![]() it is denser than the remaining h2o and thus sinks to the lesser. This "turnover" leaves a layer of warmer water most the surface, which is then cooled. All the same, if the temperature in the surface layer drops below
it is denser than the remaining h2o and thus sinks to the lesser. This "turnover" leaves a layer of warmer water most the surface, which is then cooled. All the same, if the temperature in the surface layer drops below ![]() , that water is less dense than the water below, and thus stays almost the top. Equally a result, the pond surface can freeze over. The layer of ice insulates the liquid water below it from low air temperatures. Fish and other aquatic life can survive in
, that water is less dense than the water below, and thus stays almost the top. Equally a result, the pond surface can freeze over. The layer of ice insulates the liquid water below it from low air temperatures. Fish and other aquatic life can survive in ![]() h2o below water ice, due to this unusual characteristic of water.
h2o below water ice, due to this unusual characteristic of water.
Calculating Thermal Expansion Suppose your 60.0-Fifty ![]() -gal) steel gasoline tank is full of gas that is absurd because it has just been pumped from an clandestine reservoir. Now, both the tank and the gasoline have a temperature of
-gal) steel gasoline tank is full of gas that is absurd because it has just been pumped from an clandestine reservoir. Now, both the tank and the gasoline have a temperature of ![]() How much gasoline has spilled past the fourth dimension they warm to
How much gasoline has spilled past the fourth dimension they warm to ![]() ?
?
Strategy The tank and gasoline increment in volume, but the gasoline increases more, so the amount spilled is the difference in their volume changes. We can use the equation for volume expansion to summate the modify in volume of the gasoline and of the tank. (The gasoline tank tin can be treated as solid steel.)
Solution
- Use the equation for book expansion to calculate the increase in volume of the steel tank:
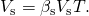
- The increment in volume of the gasoline is given by this equation:
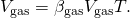
- Find the deviation in book to determine the amount spilled as
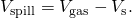
Alternatively, we can combine these three equations into a single equation. (Notation that the original volumes are equal.)
![Rendered by QuickLaTeX.com \begin{array}{cc}\hfill {V}_{\text{spill}}& =\left({\beta }_{\text{ga}}{}_{\text{s}}-{\beta }_{\text{s}}\right)V\text{Δ}T\hfill \\ & =\left[\left(950-35\right)\phantom{\rule{0.2em}{0ex}}×\phantom{\rule{0.2em}{0ex}}{10}^{-6}\text{/}\text{°}\text{C}\right]\left(60.0\phantom{\rule{0.2em}{0ex}}\text{L}\right)\left(20.0\phantom{\rule{0.2em}{0ex}}\text{°}\text{C}\right)\hfill \\ & =1.10\phantom{\rule{0.2em}{0ex}}\text{L}\text{.}\hfill \end{array}](https://opentextbc.ca/universityphysicsv2openstax/wp-content/ql-cache/quicklatex.com-38063f64b25f122b0d3e2903fedd87cc_l3.png)
Significance This corporeality is meaning, especially for a 60.0-L tank. The effect is so striking because the gasoline and steel expand quickly. The charge per unit of change in thermal properties is discussed later in this affiliate.
If you try to cap the tank tightly to prevent overflow, you will find that it leaks anyway, either around the cap or past bursting the tank. Tightly constricting the expanding gas is equivalent to compressing information technology, and both liquids and solids resist compression with extremely large forces. To avert rupturing rigid containers, these containers have air gaps, which allow them to expand and contract without stressing them.
Bank check Your Understanding Does a given reading on a gasoline gauge indicate more gasoline in cold weather condition or in hot weather, or does the temperature not thing?
The bodily amount (mass) of gasoline left in the tank when the judge hits "empty" is less in the summer than in the winter. The gasoline has the same volume equally it does in the wintertime when the "add fuel" light goes on, simply because the gasoline has expanded, there is less mass.
Thermal Stress
If you change the temperature of an object while preventing it from expanding or contracting, the object is subjected to stress that is compressive if the object would expand in the absence of constraint and tensile if it would contract. This stress resulting from temperature changes is known every bit thermal stress. It can exist quite big and tin can crusade damage.
To avert this stress, engineers may design components so they can aggrandize and contract freely. For case, in highways, gaps are deliberately left between blocks to foreclose thermal stress from developing. When no gaps can be left, engineers must consider thermal stress in their designs. Thus, the reinforcing rods in concrete are made of steel because steel'south coefficient of linear expansion is well-nigh equal to that of concrete.
To calculate the thermal stress in a rod whose ends are both fixed rigidly, nosotros can retrieve of the stress as developing in two steps. Get-go, allow the ends be free to expand (or contract) and detect the expansion (or contraction). Second, find the stress necessary to compress (or extend) the rod to its original length by the methods you studied in Static Equilibrium and Elasticity on static equilibrium and elasticity. In other words, the ![]() of the thermal expansion equals the
of the thermal expansion equals the ![]() of the rubberband distortion (except that the signs are opposite).
of the rubberband distortion (except that the signs are opposite).
Calculating Thermal Stress Concrete blocks are laid out next to each other on a highway without any infinite betwixt them, so they cannot expand. The structure coiffure did the work on a winter day when the temperature was ![]() . Find the stress in the blocks on a hot summertime day when the temperature is
. Find the stress in the blocks on a hot summertime day when the temperature is ![]() . The compressive Young'due south modulus of concrete is
. The compressive Young'due south modulus of concrete is ![]() .
.
Strategy According to the affiliate on static equilibrium and elasticity, the stress F/A is given past
![]()
where Y is the Immature's modulus of the material—concrete, in this case. In thermal expansion, ![]() We combine these ii equations by noting that the two
We combine these ii equations by noting that the two ![]() are equal, as stated above. Because nosotros are not given
are equal, as stated above. Because nosotros are not given ![]() or A, we can obtain a numerical answer merely if they both cancel out.
or A, we can obtain a numerical answer merely if they both cancel out.
Solution We substitute the thermal-expansion equation into the elasticity equation to get
![]()
and as we hoped, ![]() has canceled and A appears only in F/A, the notation for the quantity we are calculating.
has canceled and A appears only in F/A, the notation for the quantity we are calculating.
Now we need only insert the numbers:
![]()
Significance The ultimate compressive strength of concrete is ![]() then the blocks are unlikely to suspension. Withal, the ultimate shear forcefulness of physical is only
then the blocks are unlikely to suspension. Withal, the ultimate shear forcefulness of physical is only ![]() then some might chip off.
then some might chip off.
Bank check Your Understanding Two objects A and B have the aforementioned dimensions and are constrained identically. A is made of a material with a higher thermal expansion coefficient than B. If the objects are heated identically, will A experience a greater stress than B?
Non necessarily, as the thermal stress is also proportional to Young's modulus.
Summary
- Thermal expansion is the increase of the size (length, expanse, or volume) of a body due to a alter in temperature, usually a ascension. Thermal contraction is the decrease in size due to a change in temperature, usually a fall in temperature.
- Thermal stress is created when thermal expansion or wrinkle is constrained.
Conceptual Questions
Pouring cold water into hot glass or ceramic cookware can easily break it. What causes the breaking? Explicate why Pyrex®, a glass with a small coefficient of linear expansion, is less susceptible.
The cold water cools role of the inner surface, making information technology contract, while the rest remains expanded. The strain is besides great for the strength of the material. Pyrex contracts less, and then it experiences less strain.
One method of getting a tight fit, say of a metal peg in a hole in a metal block, is to industry the peg slightly larger than the hole. The peg is then inserted when at a different temperature than the block. Should the cake be hotter or colder than the peg during insertion? Explain your reply.
Does it actually help to run hot h2o over a tight metal lid on a drinking glass jar before trying to open it? Explain your answer.
In principle, the hat expands more than the jar considering metals have higher coefficients of expansion than glass. That should make unscrewing the lid easier. (In exercise, getting the lid and jar wet may brand gripping them more than difficult.)
When a cold alcohol thermometer is placed in a hot liquid, the column of alcohol goes down slightly before going up. Explicate why.
Noting the big stresses that can be caused past thermal expansion, an apprentice weapon inventor decides to utilize it to make a new kind of gun. He plans to jam a bullet confronting an aluminum rod inside a closed invar tube. When he heats the tube, the rod will expand more than the tube and a very strong force will build up. Then, by a method withal to be determined, he volition open the tube in a separate 2d and let the force of the rod launch the bullet at very high speed. What is he overlooking?
Problems
The height of the Washington Monument is measured to be 170.00 g on a day when the temperature is ![]() What will its peak be on a day when the temperature falls to
What will its peak be on a day when the temperature falls to ![]() ? Although the monument is made of limestone, presume that its coefficient of thermal expansion is the same as that of marble. Requite your reply to five pregnant figures.
? Although the monument is made of limestone, presume that its coefficient of thermal expansion is the same as that of marble. Requite your reply to five pregnant figures.
Using (Effigy) to find the coefficient of thermal expansion of marble:
![]() .
.
(Answer rounded to five significant figures to show the slight divergence in summit.)
How much taller does the Eiffel Tower become at the end of a day when the temperature has increased by ![]() Its original height is 321 chiliad and you lot can assume it is made of steel.
Its original height is 321 chiliad and you lot can assume it is made of steel.
What is the change in length of a 3.00-cm-long column of mercury if its temperature changes from ![]() to
to ![]() , assuming the mercury is constrained to a cylinder but unconstrained in length? Your answer volition prove why thermometers incorporate bulbs at the bottom instead of simple columns of liquid.
, assuming the mercury is constrained to a cylinder but unconstrained in length? Your answer volition prove why thermometers incorporate bulbs at the bottom instead of simple columns of liquid.
Using (Figure) to observe the coefficient of thermal expansion of mercury:
![]() .
.
How big an expansion gap should exist left between steel railroad rails if they may reach a maximum temperature ![]() greater than when they were laid? Their original length is 10.0 m.
greater than when they were laid? Their original length is 10.0 m.
Global warming will produce rising ocean levels partly due to melting water ice caps and partly due to the expansion of water every bit average bounding main temperatures rise. To get some thought of the size of this effect, calculate the change in length of a cavalcade of h2o one.00 km high for a temperature increase of ![]() . Presume the column is not free to expand sideways. As a model of the ocean, that is a reasonable approximation, as but parts of the ocean very shut to the surface can aggrandize sideways onto land, and only to a express caste. As some other approximation, neglect the fact that ocean warming is not uniform with depth.
. Presume the column is not free to expand sideways. As a model of the ocean, that is a reasonable approximation, as but parts of the ocean very shut to the surface can aggrandize sideways onto land, and only to a express caste. As some other approximation, neglect the fact that ocean warming is not uniform with depth.
(a) If a 500-mL glass beaker is filled to the brim with ethyl alcohol at a temperature of ![]() , how much will overflow when the alcohol's temperature reaches the room temperature of
, how much will overflow when the alcohol's temperature reaches the room temperature of ![]() ? (b) How much less water would overflow under the same atmospheric condition?
? (b) How much less water would overflow under the same atmospheric condition?
Show that ![]() by calculating the minute change in volume dV of a cube with sides of length L when the temperature changes past dT.
by calculating the minute change in volume dV of a cube with sides of length L when the temperature changes past dT.
Source: https://opentextbc.ca/universityphysicsv2openstax/chapter/thermal-expansion/
0 Response to "in this temperature range, what is the percentage change in resistance due to thermal expansion?"
Post a Comment